
by All Things Neonatal | Jun 20, 2019 | resuscitation
This post has the potential to be polarizing as sustained inflations while common as an approach after delivery in Europe has not been widely adopted in Canada and the United States. Some time ago I wrote about sustained inflations and a reader commented that I should wait for the results of the The Sustained Aeration for Infant Lungs (SAIL) trial before forming a final opinion on whether this is a good strategy or not. The previous blog post on this topic was Is It Time To Use Sustained Lung Inflation in NRP? and was followed by Is expired CO2 the key to making sustained inflation a standard in resuscitation? The first post concluded that there was a concerning trend towards more IVH in those who received sustained inflations (SI) while the second showed both a reduction in BPD and duration of mechanical ventilation with this approach. I suggested that maybe we were really onto something here and then I was asked to wait before coming to a conclusion until the SAIL trial was done. Well that day has come.
The SAIL trial
This trial led by Dr. H. Kirpalani and involving 18 NICUs in 9 countries was a big endeavour. The paper was just published and is entitled Effect of Sustained Inflations vs Intermittent Positive Pressure Ventilation on Bronchopulmonary Dysplasia or Death Among Extremely Preterm Infants The SAIL Randomized Clinical Trial. The trial compared SI of 15 seconds at a peak pressure of 20 cmH2O, followed if needed by a second SI of 15 seconds
at a peak of 25 cmH2O to traditional PPV for infants who after initial 30 seconds of CPAP required further intervention to establish breathing. These were provided via facemask or nasopharyngeal tune attached to a t-piece resuscitator. In both groups after the initial intervention standard resuscitation steps were carried out. The primary outcome was death or BPD at 36 weeks PMA. A data safety monitoring board (DSMB) was formed as well and it is this group that became very important to the conclusions of the study and led to its early termination. All infants were 23 weeks 0 days’ to 26 weeks 6 days’ GA. Before the study was terminated the final totals were 215 patients in the SI arm and 211 in the traditional PPV group.
The trial was stopped after the DSMB identified an excessive number of early deaths within 48 hours in the SI group. The findings were “11 of 16 early deaths in the sustained inflation group vs 1 of 3 in the standard
resuscitation group were considered possibly related to allocation group”. A number of these deaths occurred in the highest risk group of those born at 23-24 weeks but it was enough to stop recruitment.
With respect to the primary outcome the results showed no difference between the two approaches. In saying this however, if the study did not recruit enough patients as planned to demonstrate a difference one has to question whether the study had enough power to find a benefit.

To answer this question the authors performed a Bayesian Analysis to determine the probability that adding more patients would have led to a different conclusion. That is to determine if they would have found a difference favouring SI. In the end they found that their conclusions would not have changed. Sustained inflations in infants from 23 weeks 0 days’ to 26 weeks 6 days’ GA do not confer a benefit and may be associated with a higher likelihood of death within 48 hours of birth.
What do we do with these results?
I think this is it. I can’t see a research ethics board allowing another study at this point. This by neonatal standards was a big study given the relative scarcity of infants at these gestational ages. The fact that no difference was found in rates of death or survival with BPD for those at highest risk of these outcomes suggests to me that looking at older GA at birth will not produce different results. Sustained inflation to establish FRC and initiate respiration was a good concept backed by animal research. Moreover, clinical work out of Edmonton in recent years suggested potential benefits but with the publication of this study I suspect we will need as a neonatal community to look at other strategies to decrease rates of BPD. Concerns over increased risk of death in my opinion mean this ship has SAILed,
by All Things Neonatal | Jun 5, 2019 | resuscitation
Just about all of our preterm infants born at <29 weeks start life out the same in terms of neurological injury. There are of course some infants who may have suffered ischemic injury in utero or an IVH but most are born with their story yet to be told. I think intuitively we have known for some time that the way we resuscitate matters. Establishing an FRC by inflating the lungs of these infants after delivery is a must but as the saying goes the devil is in the details.
The Edmonton group led by Dr. Schmolzer has had several papers examined in these blogs and on this occasion I am reviewing an important paper that really is a follow-up study to a previous one looking at the impact of high tidal volume delivery after birth. I have written on this previous paper before in It’s possibile! Resuscitation with volume ventilation after delivery. On this occasion the authors have published the following paper; Impact of delivered tidal volume on the occurrence of intraventricular haemorrhage in preterm infants during positive pressure ventilation in the delivery room.This observational study had a simple enough premise. Will the use of Vt > 6 mL/kg in infants given PPV for at least two minutes lead to worse rates of IVH? All infants were < 29 weeks and if they had chest compressions or epinephrine were excluded. All infants were treated equally in terms of delayed cord clamping and antenatal steroid provision. Ventilation was done with a t-piece resuscitator and Vt measured with an NM3 monitor connected to the face mask. First ultrasounds were done for all at 3 days of age.
What did the authors find?
One hundred and sixty five infants comprised this cohort. Overall, 124 (75%) infants were in the high volume group compared to 41 (25%) with a mean VT<6 mL/kg. Median Vt were 5.3 (4.6-5.7) ml/kg for the low group and 8.7
(7.3-10.6) mL/kg which were significantly different.
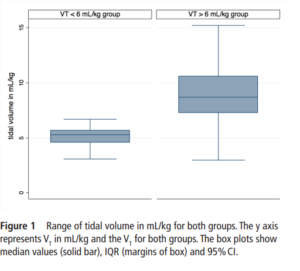
 When looking at the rates of IVH and the severity of those affected the results are striking.
When looking at the rates of IVH and the severity of those affected the results are striking.
IVH in the high VT group was diagnosed in 63 (51%) infants compared with 5 (13%) infants in the normal VT group (P=0.008).Severe IVH (grade III or IV) developed in 33/124 (27%) infants in the high VT group and 2/41 (6%) in the normal VT group (P=0.01)
Hydrocephalus, following IVH developed in 7/49 (14%) and 2/16 (13%) in the >6 mL/kg and <6 mL/kg VT groups. Looking at other factors that could affect the outcome of interest the authors noted the following physiologic findings. Oxygen saturations were lower in the low volume group at 6, 13 and 14 min after birth while tissue oxygenation as measured by NIRS was similarly lower at 7,8 and 25 min after birth (P<0.001). Conversely, heart rate was significantly lower in the VT>6 mL/kg group at 5, 20 and 25 min after birth (P<0.001). Fraction of inspired oxygen was similar in both groups within the first 30 min. Systolic, diastolic and mean blood pressure was similar between the groups. What these results say to me is that despite having lower oxygen saturations and cerebral oxygen saturation at various time points in the first 25 minutes of life the infants seem to be better off given that HR was lower in those given higher volumes despite similar FiO2. Rates of volume support after admission were slightly higher in the high volume group but inotrope usage appears to be not significantly different. Prophylactic indomethacin was used equally in the two cohorts.
Thoughts for the future
Once a preterm infant is admitted to the NICU we start volume targeted ventilation from the start. In the delivery room we may think that we do the same by putting such infants on a volume guarantee mode after intubation but the period prior to that is generally done with a bag and mask. Whether you use a t-piece resuscitator or an anesthesia bag or even a self inflating bag, you are using a pressure and hoping not to overdistend the alveoli. What I think this study demonstrates similar to the previous work by this group is that there is another way. If we are so concerned about volutrauma in the NICU then why should we feel any differently about the first few minutes of life. Impairment of venous return from the head is likely to account for a higher risk of IVH and while a larger study may be wished for, the results here are fairly dramatic. Turning the question around, one could ask if there is harm in using a volume targeted strategy in the delivery room? I think we would be hard pressed to say that keeping the volumes under 6 mL/kg is a bad idea. The challenge as I see it now is whether we rig up devices to accomplish this or do the large medical equipment providers develop an all in one system to accomplish this? I think the time has come to do so and will be first in line to try it out if there is a possibility to do a trial.

by All Things Neonatal | May 23, 2019 | resuscitation
We have all been there. After an uneventful pregnancy a mother presents to the labour floor in active labour. The families world is turned upside down and she goes on to deliver an infant at 27 weeks. If the infant is well and receives minimal resuscitation and is on CPAP we provide reassurance and have an optimistic tone. If however their infant is born apneic and bradycardic and goes on to receive chest compressions +/- epinephrine what do we tell them? This infant obviously is much sicker after delivery and when the family asks you “will my baby be ok?” what do you tell them? It is a human tendency to want to reassure and support but if they ask you what the chances are of a good outcome it has always been hard to estimate. What many of us would default to is making an assumption that the need for CPR at a time when the brain is so fragile may lead to bleeding or ischemia would lead to worse outcomes. You would mostly be right. One study by Finer et al entitled Intact survival in extremely low birth weight infants after delivery room resuscitation.demonstrated that survival for infants under 750g was better if they had a history of CPR after delivery. The thought here is that more aggressive resusctiation might be responsible for the better outcome by I would presume establishing adequate circulation sooner even if the neonates did not appear to need it immediately.
The Canadian Neonatal Network
In Canada we are fortunate to have a wonderful network called the Canadian Neonatal Network. So many questions have been answered by examining this rich database of NICUs across the county. Using this database the following paper was just published by Dr. A. Lodha and others; Extensive cardiopulmonary resuscitation of preterm neonates at birth and mortality and developmental outcomes. The paper asked a very specific and answerable question from the database. For infants born at <29 weeks gestational age who require extensive resuscitation (chest compressions, epinephrine or both) what is the likelihood of survival and/or neurodevelopmental impairment (NDI) at 18-24 months of age vs those that did not undergo such resuscitation? For NDI, the authors used a fairly standard definition as “any cerebral palsy (GMFCS�1), Bayley-III score <85 on one or more of the cognitive, motor or language composite scores, sensorineural or mixed hearing impairment or unilateral or bilateral visual impairment.” Their secondary outcomes were significant neurodevelopmental impairment (sNDI), mortality, a Bayley-III score of <85 on any one of the components (cognitive, language, motor), sensorineural or mixed hearing loss,or visual impairment.sNDI was defined as the presence of one or more of the following: cerebral palsy with GMFCS 3, Bayley-III cognitive, language or motor composite score <70, hearing impairment requiring hearing aids or cochlear implant, or bilateral visual impairment”
What did they discover?
It is a fortunate thing that the database is so large as when you are looking at something like this the number of infants requiring extensive resuscitation is expected to be small. The authors collected data from January 1, 2010 and September 30, 2011 and had a total number of infants born at less than 29 weeks of 2760. After excluding those with congenital anomalies and those who were born moribund they were left with 2587. From these 80% had follow-up data and when applying the final filter of extensive resuscitation they were left with 190 (9.2%) who received delivery room CPR (DR-CPR) vs 1545 who did not receive this.
Before delving into the actual outcomes it is important to note that neonates who did not receive DR-CPR were more likely to be born to mothers with hypertension and to have received antenatal steroids (89 vs 75%). With these caveats it is pretty clear that as opposed to the earlier study showing better outcomes after DR-CPR this was not the case here.

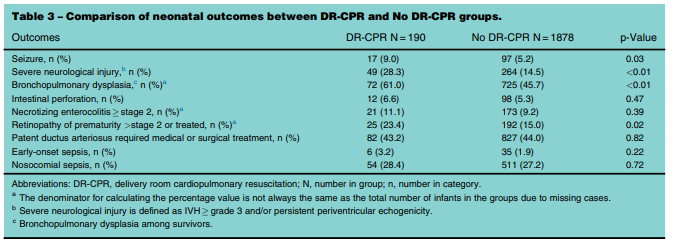
 The results are interesting in that it is pretty clear that receiving DR-CPR is not without consequence (higher rate of seizures, severe neurological injury, BPD). Looking at the longer term outcomes though is where things get a little more interesting. Mortality and mortality or neurodevelopmental impairment are statistically significant with respect to increased risk. When you take out NDI alone however the CI crosses one and is no longer significant. Neither is CP for that matter with the only statistically significant difference being the Bayley-III Motor composite score <85. The fact that only this one finding came out as significant at least to me raises the possibility that this could have been brought about by chance. It would seem that while these infants are at risk of some serious issues their brains in the long run may be benefiting for the neurological plasticity that we know these infants have.
The results are interesting in that it is pretty clear that receiving DR-CPR is not without consequence (higher rate of seizures, severe neurological injury, BPD). Looking at the longer term outcomes though is where things get a little more interesting. Mortality and mortality or neurodevelopmental impairment are statistically significant with respect to increased risk. When you take out NDI alone however the CI crosses one and is no longer significant. Neither is CP for that matter with the only statistically significant difference being the Bayley-III Motor composite score <85. The fact that only this one finding came out as significant at least to me raises the possibility that this could have been brought about by chance. It would seem that while these infants are at risk of some serious issues their brains in the long run may be benefiting for the neurological plasticity that we know these infants have.
The study is remarkable to me in that an infant can have such a difficult start to life yet hope may remain even after dealing with some of the trials and tribulations of the NICU. Parents may need to wade through the troubling times of seizures, long term ventilation and CPAP and then onto a diagosis of BPD but their brains may be ok after all. This is one of the reasons I love what I do!

by All Things Neonatal | Jan 3, 2019 | resuscitation
In July 2016 I published a blog post No more intubating for meconium? Not quite. In this post I highlighted the recent recommendations to modify the approach to the non vigorous infant born through meconium. The traditional approach of electively intubating such infants for tracheal suctioning before beginning PPV was replaced by provision of PPV first. The rationale here was that delaying the establishment of ventilation while trying to intubate for most situations was more risky than just trying to establish a functional residual capacity (FRC). The naysayers pointed out that while this recommendation is possibly warranted for less experienced intubators, perhaps in the hands of those with more skill, tracheal suctioning would be the better option if it could on average be done quickly.
It has been over two years since that recommendation and change in practice. Isn’t it about time someone looked at whether or not this was a good thing to do?
A Comparison of Two Time Periods
Chiruvolu A et al published Delivery Room Management of Meconium-Stained Newborns and Respiratory Support in this month’s Pediatrics. In this paper the authors compared 4 hospitals with a retrospective period of one year before the NRP changes (October 1, 2015, to September 30, 2016) to a one year prospective period (October 1, 2016, to September 30, 2017) after implementation of the new guidelines. In the retrospective cohort there were 11163 mothers delivered at ≥35 weeks’ gestation. Meconium stained amniotic fluid (MSAF) was present in 1303 (12%) deliveries with 130 (10%) of newborns who were nonvigorous. During the prospective time period, a total of 10 717 mothers delivered at ≥35 weeks’ gestation. MSAF was noted in 1282 (12%) deliveries, yielding 101 (8%) newborns who were nonvigorous. Therefore the study compared these 130 newborns in the retrospective cohort to the 101 in the prospective time period. The authors note that aside from the approach to MSAF there were no changes in care during this time in the delivery room.
A few differences exist though in the cohorts that are worth mentioning that were statistically significant. Firstly, the incidence of preterm and post-term infants were both higher in the prospective cohort (both 6% vs 1%). Secondly, the incidence of fetal distress was higher in the prospective cohort 57% vs 43%. All of these factors would tend to favour the retrospective cohort doing better than the prospective and so the authors in their results controlled for these differences. Not surprisingly the rate of intubation in the retrospective group was 70% vs 2% in the prospective arm.
What were the results?
The results shown in table 3 in terms of the Odds ratios have been adjusted for the aforementioned differences of preterm post-term and fetal distress. There are several things here worth noting.  The risk of admission was significantly higher for respiratory distress.
The risk of admission was significantly higher for respiratory distress.
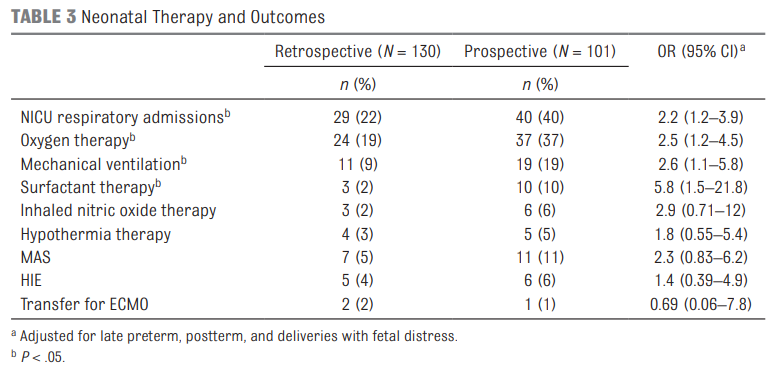 Oxygen needs and mechanical ventilation along with surfactant therapy were also notably higher. One things that showed no difference at all was the mean apgar score at 1 and 5 minutes. This is an interesting finding given the hypothesis that drove the change in practice. If establishing an FRC is the goal of the intervention to provide earlier PPV then shouldn’t the retrospective group have worse apgars due to less effective resuscitation? Maybe or maybe not. This really depends on the staff in the resuscitation room at the 4 hospitals. It might be that the staff were quite skilled so the intubations may have gone smoothly with minimal reductions in FRC compared to the prospective group. What would this study look like if done in a centre with less experienced people capable of intubation.
Oxygen needs and mechanical ventilation along with surfactant therapy were also notably higher. One things that showed no difference at all was the mean apgar score at 1 and 5 minutes. This is an interesting finding given the hypothesis that drove the change in practice. If establishing an FRC is the goal of the intervention to provide earlier PPV then shouldn’t the retrospective group have worse apgars due to less effective resuscitation? Maybe or maybe not. This really depends on the staff in the resuscitation room at the 4 hospitals. It might be that the staff were quite skilled so the intubations may have gone smoothly with minimal reductions in FRC compared to the prospective group. What would this study look like if done in a centre with less experienced people capable of intubation.
Also interesting in this study is that when isolating comparisons to those admitted to the NICU and those specifically diagnosed with MAS there were no differences between groups for such outcomes as length of stay, oxygen therapy, mechanical ventilation (MV) or days of MV. Given that the group sizes though were quite small (7 and 11 for MAS) we do have to take this data with a grain of salt as it really is too small to make any certain conclusions. A larger study would need to be done looking at these types of outcomes to really get a better handle on whether the approach to MSAF matters to these individual outcomes.
Final Thoughts
What this study does for me is raise an eyebrow. The change in practice does not seem to yield “better babies”. Secondly what we do see even when controlling for differences that would affect hospital admissions for respiratory distress is an increase in admission rate. In times when beds are becoming increasingly precious as census for many units swell one has to ask whether this approach is truly the better way to go. Perhaps it was wrong for the NRP to declare that for all practitioners it is best to provide PPV rather than intubate. This may have been too simplistic. If you have experienced intubators perhaps it would be best to continue to intubate first in this setting rather than provide PPV. What this study does is certainly raise questions and begs for a larger study to be done to determine whether these results can be replicated. If they are then I suspect the NRP may be headed down a different path for recommendations yet again.
by All Things Neonatal | Nov 7, 2018 | resuscitation
Much has been written on the topic of cord clamping. There is delayed cord clamping of course but institutions differ on the recommended duration. Thirty seconds, one minute or two or even sometimes three have been advocated for but in the end do we really know what is right? Then there is also the possibility of cord milking which has gained variable traction over the years. A recent review was published here.
Take the Guessing Out of the Picture?
Up until the time of birth there is very little pulmonary blood flow. Typically, about 10% of the cardiac output passes through the lungs and the remained either moves up the ascending aorta or bypasses the lungs via the ductus arteriosus. After birth as the lung expands, pulmonary vascular resistance rapidly decreases allowing cardiac output to take on the familiar pattern which we all live with. Blood returning from the systemic venous circulation no longer bypasses the lung but instead flows through pulmonary capillaries picking up oxygen along the way. One can imagine then that if a baby is born and the cord is clamped right away, blood returning from the systemic circulation continues to bypass the lung which could lead to hypoxemia and reflexive bradycardia. This has been described previously by Blank et al in their paper Haemodynamic effects of umbilical cord milking in premature sheep during the neonatal transition.
A group of researchers from the Netherlands published a very interesting paper Physiological-based cord clamping in preterm infants using a new purpose-built resuscitation table: a feasibility study this month. The study centres around a resuscitation table called the Concord that is brought to the mother for resuscitation after birth.  The intervention here was applied to infants 26 to 35 weeks gestational age.
The intervention here was applied to infants 26 to 35 weeks gestational age.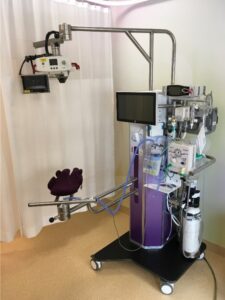
The cord was clamped after each of the following was achieved for an infant indicating successful transition with opening of the lung and establishment of an FRC.
1. Establishment of adequate breathing (average tidal volume ≥4 mL/kg) on CPAP. They used a mask capable of measuring expired tidal volumes.
2. HR above 100 bpm
3. SpO2 above 25th percentile using FiO2 <0.4
In this way, the cord was only clamped once the baby appeared to have physiologically made the transition from dependence on umbilical cord blood flow to ventilation perfusion matching in the lung. Although 82 mothers consented only 37 preterm infants were included in the end. Exclusion criteria were signs of placental abruption or placenta praevia, signs of severe fetal distress determined by the clinician and the necessity for an emergency caesarean section ordered to be executed within 15 min. This really was a proof of concept study but the results are definitely worth looking at.
How Did These Babies Do?
There are many interesting findings from this study. The mean time of cord clamping was 4 minutes and 23 seconds (IQR 3:00 – 5:11). Heart rate was 113 (81–143) and 144 (129–155) bpm at 1 min and 5 min
after birth. Only one patient developed bradycardia to <60 BPM but this was during a mask readjustement. The main issue noted as far as adverse events was hypothermia with a mean temperature of 36.0 degrees at NICU admission. Almost 50% of infants had a temperature below 36 degrees. Although the authors clearly indicate that they took measures to prevent heat loss it would appear that this could be improved upon!
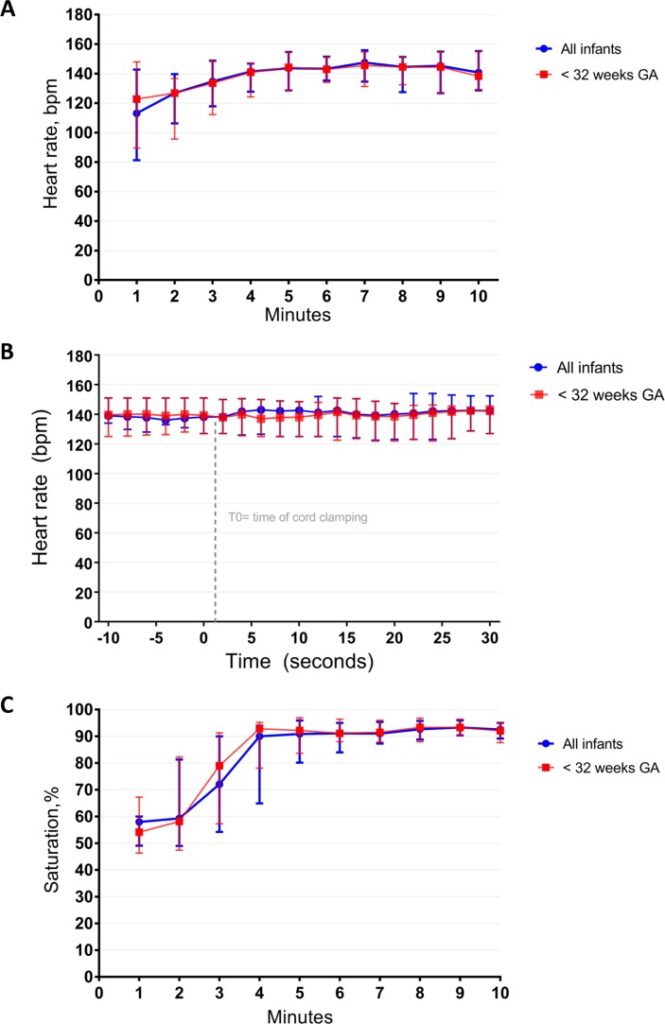
What stands out most to me is the lengthy duration of cord clamping. This study which used a physiologic basis to determine when to clamp a cord has demonstrated that even at 1 minute of waiting that is likely only 1/4 of the time needed to wait for lung expansion to occur to any significant degree. I can’t help but wonder how many of the patients we see between 26-35 weeks who have a low heart rate after delivery might have a higher heart rate if they were given far more time than we currently provide for cord clamping.
I can also see why cord milking may be less effective. Yes, you will increase circulating blood volume which may help with hemodynamic stability but perhaps the key here is lung expansion. You can transfuse all the blood you want but if it has nowhere to go just how effective is it?
As we do more work in this area I have to believe that as a Neonatal community we need to prepare ourselves for the coming of the longer delay for cord clamping. Do we need to really have the “Concord” in every delivery or perhaps it is time to truly look at durations of 3-4 minutes before the team clamps the cord.
Stay tuned!





 When looking at the rates of IVH and the severity of those affected the results are striking.
When looking at the rates of IVH and the severity of those affected the results are striking.




 The risk of admission was significantly higher for respiratory distress.
The risk of admission was significantly higher for respiratory distress. Oxygen needs and mechanical ventilation along with surfactant therapy were also notably higher. One things that showed no difference at all was the mean apgar score at 1 and 5 minutes. This is an interesting finding given the hypothesis that drove the change in practice. If establishing an FRC is the goal of the intervention to provide earlier PPV then shouldn’t the retrospective group have worse apgars due to less effective resuscitation? Maybe or maybe not. This really depends on the staff in the resuscitation room at the 4 hospitals. It might be that the staff were quite skilled so the intubations may have gone smoothly with minimal reductions in FRC compared to the prospective group. What would this study look like if done in a centre with less experienced people capable of intubation.
Oxygen needs and mechanical ventilation along with surfactant therapy were also notably higher. One things that showed no difference at all was the mean apgar score at 1 and 5 minutes. This is an interesting finding given the hypothesis that drove the change in practice. If establishing an FRC is the goal of the intervention to provide earlier PPV then shouldn’t the retrospective group have worse apgars due to less effective resuscitation? Maybe or maybe not. This really depends on the staff in the resuscitation room at the 4 hospitals. It might be that the staff were quite skilled so the intubations may have gone smoothly with minimal reductions in FRC compared to the prospective group. What would this study look like if done in a centre with less experienced people capable of intubation.
 The intervention here was applied to infants 26 to 35 weeks gestational age.
The intervention here was applied to infants 26 to 35 weeks gestational age.
