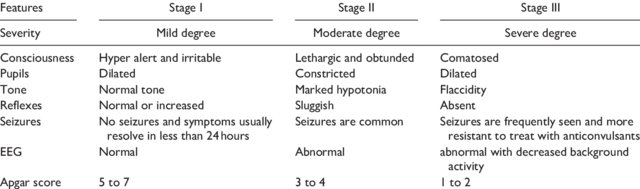
Hypoxic ischemic encephalopathy is a very scary condition for both families and health care providers. In my career as a Neonatologist one of the greatest accomplishments has been the recognition that 72 hours of moderate hypothermia can make a big difference to the outcomes of such children. In days gone by our best estimates of outcome relied on Sarnat staging of HIE.
Since the cooling approach was adopted widely however I have relied more on a wait and see approach when advising families on what to expect. On some occasions, is spite of cooling babies go on to develop significant cerebral palsy but in other cases babies who one would have predicted would have dismal outcomes have done quite well. Our best estimates at the moment are that cooling for HIE reduces the risk of death or moderate to severe disability by about 25% with a confidence interval of 17 – 32% around that estimate.
Why would pCO2 matter?
Carbon dioxide has a role to play in outcome and has been the subject of several papers. The theoretical point is that very low carbon dioxide levels lead to vasoconstriction of blood vessels. When it comes to HIE one would be quite worried about vasoconstriction of blood vessels such as the carotids carrying oxygenated blood to an injured brain. Once injured the brain is not going to tolerate further oxygen deprivation and in particular those areas that are teetering on the edge of survival could be tipped the wrong way if further hypoxia is experienced.
Another reason why CO2 matters is due to something called the Bohr effect. For those of you who are scratching your heads and recollecting this term from your training it has to do with the influence of pCO2 on hemoglobin oxygen saturation. The relationship is represented by the following figure.
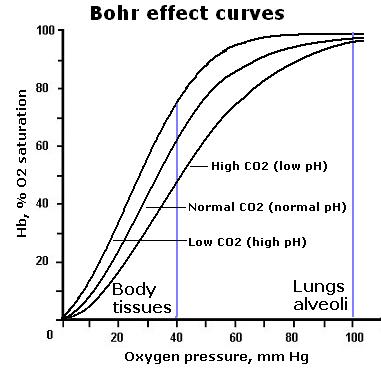
In the presence of declining pCO2 there is a shift of the oxygen dissociation curve to the left. This means for that as PCO2 declines more of the circulating oxygen will be bound to hemoglobin. In most cases you want your hemoglobin to be great at carrying oxygen but when your tissues are starved of oxygen and injured that is not what you want. You want a selfless hemoglobin molecule that is more than happy to release its oxygen to the tissue. That is not what you get as pCO2 drops.
Why would pCO2 be low at all?
There are a few reasons for this. The first is that many infants born after an asphyxial event have a metabolic acidosis. Our bodies naturally like to maintain a normal pH. In order to do so if your HCO3 in the blood is low you need to blow off CO2 to compensate. The hypocarbia in this case is compensatory but the body in so doing could make matters worse for the brain.
The second reason has to do with both injured tissue and that which is cooled. As metabolic rate decreases the amount of CO2 generated will drop. If you remember the Krebs cycle (shudder) there is a fair bit of CO2 generated from aerobic (oxygen rich) metabolism. If this is reduced so to will the production of CO2. As cooling serves to reduce metabolic rate so the CO2 production would be expected to decrease.
So does it really matter?
The reason for all this preamble is that a “mini-systematic review” has found the CO2 matters to outcome. The review is entitled Hypocarbia is associated with adverse outcomes in hypoxic ischaemic encephalopathy (HIE) and included 9 studies on influence of pCO2 on outcome. Before we look at the results it is important to acknowledge that all of the included studies were retrospective so methodology in each study is not standardized. How one even defines severe hypocarbia varied from <20 mmHg to anything under 35 mm Hg. The other issue is that each study looked at a different period of exposure from the effect of a couple hours to the effect over the first three days of life. The included infants were all cooled so it gives us at least an idea of the effect in a modern cohort of cooled infants.
The summary of the results was that CO2 mattered. As little as a couple hours of very low CO2 levels were found to be associated with adverse outcomes.
The problem of course is the chicken and the egg argument. The most severe hypocarbia might be seen in those with the worst metabolic acidosis. As mentioned above the response to metabolic acidosis is to blow off CO2. Therefore, the worse the metabolic acidosis the greater that respiratory drive.
Strategies to control the pCO2 of course exist. In the presence of a critically low pCO2 one can intubate and control ventilation through sedation and paralysis. This can lead to other issues though as if you normalize the pCO2 in the presence of a significant metabolic acidosis the pH is likely to take a nosedive. The myocardium as it turns out doesn’t like low pH and in fact cardiac output in animal models begins to decrease the closer you get to a pH of 7 and becomes significantly worse as you go beyond that point.
At best then I think one can aim for converting severe hypocarbia to moderate until the HCO3 begins to recover. Based on theoretical issues of oxygen delivery to tissues and cerebral vasoconstriction, notwithstanding the retrospective nature of this review it does make sense to me that there would be a link between severe hypocarbia and outcome. We will likely never see an RCT targeting normalization of pCO2 vs tolerance of hypocarbia in this population so for the purists out there that don’t like this type of retrospective analysis I suspect outside of an animal model this is as good as its going to get.
Maybe avoiding anything with the word severe attached to it though is sensible when it comes to this population.


Great discussion on CO2, specially on infants with HIE. Yes, we are concern in these cases with low PaCO2 such as increase intracranial pressure, metabolic acidosis and metabolic derangements. Do we control the PaCO2 at expense of pH? Do we control PaCO2 thru respiratory manipulation and increased other risks as pulmonary and cardiovascular side effects? Great idea for several clinical or animals trials.
We cooled but did not permit hypocarbia.