
by All Things Neonatal | Dec 26, 2020 | Neonatal, Parents Corner, Unit design
Some time ago Nick Hall from Graham’s Foundation posted the following question on Linkedin:
Private room vs open bay for the NICU. Can always get a quote from a parent saying it is great but….? At what cost? Impact on staff? Is parent time in those NICUs greater now? Other alternatives?
Included in the post was an article discussing the benefits of such a design. Below I will look at the benefits and risks and conclude with an answer to his last question.
The NICUs of the 1970s through late 1990s have been described as “barn like” or “open concept” but in recent years the belief that single patient rooms (SPR) would offer greater benefit to infants led to the adoption of such a unit design across North America. The imagined benefits would be related to improved parent comfort, creating a desire for families to spend more time with their children. As we move to a “family centred” approach to care, a key goal of all units should be to make their families as comfortable and stress free as possible in order to have a positive experience.
Detractors meanwhile, speak of concern regarding isolation of such infants when families do not visit and moreover a risk that such infants deprived of sensory experience will have impaired development. Last year a paper was published that did not help quell such fears; Alterations in Brain Structure and Neurodevelopmental Outcome in Preterm Infants Hospitalized in Different Neonatal Intensive Care Unit Environments (full article in link). This study which compared infants cared for in SPR to an open unit (the hospital in this study had a mixture of both in their NICU) found a worrisome finding at 2 year follow-up in that the infants in SPR had lower scores on language and a trend towards lower motor scores as well. Additionally, partly explaining such findings may have been differences noted at term equivalent age in both the structure and activity of the children’s brains compared to those cared for in an open environment. We were starting construction on a new NICU at the time this paper was published and I can tell you the findings sent shockwaves through our hospital as many wondered whether this was the right decision.
Devil Is in The Details
Looking further into this study, the urban population bore little resemblance to our own. In our hospital all women are taught how to perform skin to skin care and the majority of our mothers spend a great deal of time with their infants. To see how successful have a look at our recent Kangaroo Care drive results! The families in this study however the average hours per week of parent visitation over the length of stay ranged from 1.8-104 hours with a mean of 19+/- 19 hours. The average number of days held per week over the length of stay was 0-6 days with a mean of 2.4 +/-1.5 days. The average number of days held skin-to-skin over the length of stay ranged from 0-4 days, with a mean of 0.7 +/- 0.9 days. In short they were hardly there.
Second Study Finds The Opposite
Later on in 2014 a second study on this subject was published; Single-family room care and neurobehavioral and medical outcomes in preterm infants. Infants < 1500g who were admitted to an NICU between 2008 and 2012 were compared with respect to medical and neurobehavioral outcomes at discharge. Participants included 151 infants in an open-bay NICU and 252 infants after transition to a SPR NICU.
Statistically significant results (all Ps ≤.05) showed that infants in the SPR NICU weighed more at discharge, had a greater rate of weight gain, required fewer medical procedures, had a lower gestational age at full enteral feed and less sepsis, showed better attention, less physiologic stress, less hypertonicity, less lethargy, and less pain. Nurses reported a more positive work environment and attitudes in the SPR NICU.
This study in fact demonstrated greater maternal involvement in a SPR with improvement in outcomes across the board. It would seem then that in a SPR environment, provided there is enough family visitation and involvement this model truly is superior to the open concept. Furthermore despite concerns by some nurses that the loss of line of sight to their patients will make for a more stressful working environment this does not seem to be the case.
What About Families Who Cannot or Simply Aren’t Visiting Frequently?
The reality is that there are many reasons for parents to be absent for long periods during their newborns stay. Having a home outside of the city with other children to care for, work obligations, or loss of custody and abandonment due to apprehension are just some of these reasons. In our hospital, at least 15-20% of all patients admitted are from outside Winnipeg. The evidence as I see it supports the move to a SPR but what do we do for those children who need more visitation? The solution is a cuddler program.
 In our new hospital we are grateful for the generosity of our Children’s Hospital Foundation who secured a donor to pay for a coordinator of such a program. The veteran parent who is leading this program ensures that no infant goes beyond a set period of time without feeling the touch or hearing the sound of a voice. Such a program is in fact already in place at our other tertiary hospital and was featured in a lovely article attached here. Taking all the information together that is out there I think that if we can provide the necessary stimulation from both touch and auditory stimuli as well we can provide these infants with the developmental needs that each of them requires.
In our new hospital we are grateful for the generosity of our Children’s Hospital Foundation who secured a donor to pay for a coordinator of such a program. The veteran parent who is leading this program ensures that no infant goes beyond a set period of time without feeling the touch or hearing the sound of a voice. Such a program is in fact already in place at our other tertiary hospital and was featured in a lovely article attached here. Taking all the information together that is out there I think that if we can provide the necessary stimulation from both touch and auditory stimuli as well we can provide these infants with the developmental needs that each of them requires.
The SPR is the right design in my mind for families with many benefits that spring forth in such an environment. This need not be a win-lose scenario for your hospital. Do not underestimate the power of a cuddler and don’t hesitate to seek support to initiate such a program. It could mean the difference from going from good to great!

by All Things Neonatal | Sep 6, 2018 | Neonatal, Neonatology, newborn, preemie
It has been a few months now that I have been serving as Chair of the Fetus and Newborn Committee for the Canadian Pediatric Society. Certain statements that we release resonate strongly with me and the one just released this week is certainly one of them. Guidelines for vitamin K prophylaxis in newborns is an important statement about a condition that thankfully so few people ever experience. To read the statement on the CPS website click here.
Similar story to vaccinations
Prior to the American Academy of Pediatrics in 1961 proclaiming that all newborns should receive IM Vitamin K at birth the incidence of Vitamin K deficient bleeding was 0.25 – 1.7%. Think about that for a moment. A new parent could expect that 1/100 babies roughly might have intestinal bleeding or worse an intracranial hemorrhage due to an insufficient amount of vitamin K levels in the newborn. The types of bleeding could be categorized into three different time epochs. Early onset (occurring in the first 24 hours post-birth), classic (occurring at days 2 to 7) and late onset (at 2 to 12 weeks and up to 6 months of age).
With a rate that high detractors of providing Vitamin K at birth would say “why should we give it; I haven’t heard of any baby getting such bleeding?” Looking at it another way though, why don’t you see congenital rubella or kids with measles much these days? It’s due to vaccination. Thankfully as a Neonatologist, I don’t see Vitamin K deficient bleeding since most parents provide Vitamin K to their babies at birth. If you went back to the era prior to 1961 when widespread supplementation of Vitamin K began in the US, I imagine it would not have been too uncommon to hear about a baby who had bleeding issues after birth. Just because we don’t hear about German Measles much anymore doesn’t mean the virus causing it doesn’t still exist!
How Effective is Vitamin K?
How effective is Vitamin K administration at birth in preventing hemorrhagic disease of the newborn (HDNB)? Studies estimate an incidence of 0.25 per 100000 live births or 1 in 400000 babies vs the 1/100 risk without any vitamin K. That is one effective intervention! At this point I would ask those families that are still concerned about giving Vitamin K to their infants if this is a risk they can accept? If they refuse Vitamin K and there is a significant bleed how will they react?
The Change in this CPS Statement From the Past
In the last statement on Vitamin K, the authors suggested that the oral route was a reasonable option. Instead of giving 1 mg of Vitamin K IM one would dose it as 2 mg orally and then repeat at 2-4 weeks and then 6-8 weeks. In looking at the effectiveness though it is worth noting that while we can assure that families will get the first dose, as with any medication that needs repeat dosing there is the risk of forgetfulness leading to missed dosing down the road. In fact when the authors looked at the risk of late HDNB they found the following “The relative risk for VKDB, when comparing PO versus IM vitamin K administration in these two studies, was 28.75 (95% CI 1.64 to 503.45) and 5.97 (95% CI 0.54 to 65.82), respectively [19][20].”
The outcome of course remains rare but the risk based on two studies was almost 30 times higher than if IM dosing was given.
On this basis IM is recommended.
Having said all this I recognize that despite all this information, some families will choose for a number of reasons to still opt for the oral dose. As the statement suggests we need to encourage such use when a family refuses IM vitamin K. The 30 fold risk compared to IM administration is magnitudes lower than the approximate 1/100 risk of giving nothing at all!
In the end I believe that one case of intracranial hemorrhage from inadequate vitamin K is too much. This one vitamin indeed could save a life.

by All Things Neonatal | Aug 15, 2018 | intubation, Neonatal, Neonatology, technology
The modern NICU is one that is full of patients on CPAP these days. As I have mentioned before, the opportunity to intubate is therefore becoming more and more rare is non-invasive pressure support becomes the mainstay of therapy. Even for those with established skills in placing an endotracheal tube, the number of times one gets to do this per year is certainly becoming fewer and fewer. Coming to the rescue is the promise of easier intubations by being able to visualize an airway on a screen using a video laryngoscope. The advantage to the user is that anyone who is watching can give you some great tips and armed with this knowledge you may be better able to determine how to adjust your approach.
For those of you who have followed the blog for some time, you will recall this is not the first time video laryngoscopy has come up. I have spoken about this before in Can Video Laryngoscopy Improve Trainee Success in Intubation. In that piece, the case was made that training residents how to intubate using a video laryngoscope (VL) improves their success rate. An additional question that one might ask though has to do with the quality of the intubation. What if you can place a tube using a video laryngoscope but the patient suffers in some way from having that piece of equipment in the mouth? Lucky for us some researchers from the Children’s Hospital of Philadelphia have completed a study that can help answer this additional question.
Video Laryngoscopy may work but does it cause more harm than good?
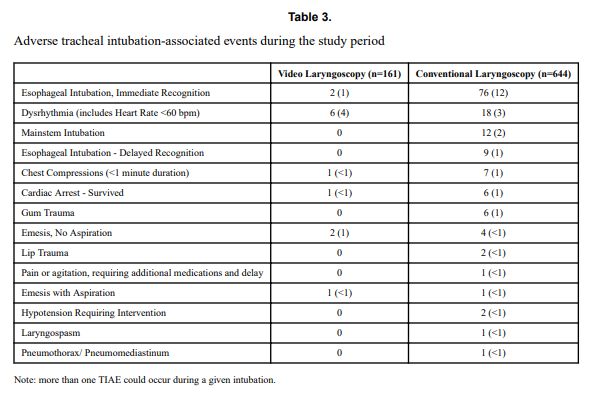
Using a video laryngoscope requires purchasing one first and they aren’t necessarily cheap. If they were to provide a better patient experience though the added cost might well be worth it. Pouppirt NR et al published Association Between Video Laryngoscopy and Adverse Tracheal Intubation-Associated Events in the Neonatal Care Unit. This study was a retrospective comparison of two groups; one having an intubation performed with a VL (n=161 or 20% of the group) and the other with a standard laryngoscope (644 or 80% of the group). The study relied on the use of the National Emergency Airway Registry for Neonates (NEAR4NEOs), which records all intubations from a number of centres using an online database and allows for analysis of many different aspects of intubations in neonates. In this case the data utilized though was from their centre only to minimize variation in premedication and practitioner experience.
Tracheal intubation adverse events (TIAEs) were subdivided into severe (cardiac arrest, esophageal intubation with delayed recognition, emesis with witnessed aspiration, hypotension requiring intervention (fluid and/or vasopressors), laryngospasm, malignant hyperthermia, pneumothorax/pneumomediastinum, or direct airway injury) vs non-severe (mainstem bronchial intubation, esophageal intubation with immediate recognition, emesis without aspiration, hypertension requiring therapy, epistaxis, lip trauma, gum or oral trauma, dysrhythmia, and pain and/or agitation requiring additional medication and causing a delay in intubation.
Looking at the patient characteristics and outcomes, some interesting findings emerge.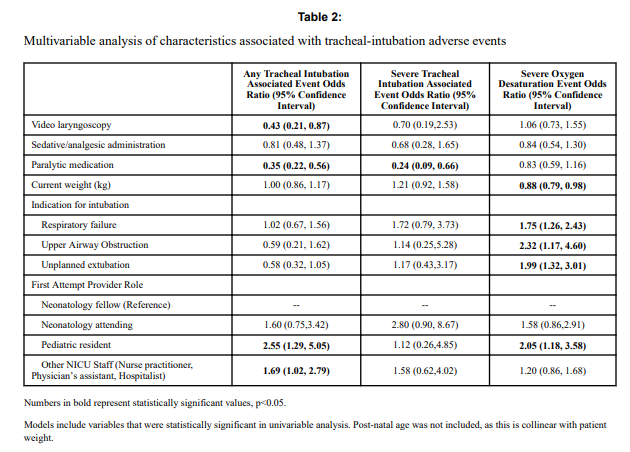

Patients who had the use of the VL were older and weighed more. They were more likely to have the VL used for airway obstruction than respiratory failure and importantly were also more likely to receive sedation/analgesia and paralysis. These researchers have also recently shown that the use of paralysis is associated with less TIAEs so one needs to bear this in mind when looking at the rates of TIAEs. There were a statistically significant difference in TIAEs of any type of 6% in the VL group to 19% in the traditional laryngoscopy arm but severe TIAEs showed not difference.
Given that several of the baseline characteristics might play a role in explaining why VL seemed superior in terms of minimizing risk of TIAEs by two thirds, the authors performed a multivariable analysis in which they took all factors that were different into account and then looked to see if there was still an effect of the VL despite these seemingly important differences. Interestingly, use of VL showed an Odds ratio of 0.43 (0.21,0.87 95% CI) in spite of these differences.
What does it mean?
Video laryngoscopy appears to make a difference to reducing the risk on TIAEs as an independent factor. The most common TIAE was esophageal intubation at 10% and reducing that is a good thing as it leads to fewer intubation attempts. This was also sen as the first attempt success was 63% in the VL group vs 44% in the other.
Now we need to acknowledge that this was not a randomized controlled trial so it could indeed be that there are other factors that the authors have not identified that led to improvements in TIAEs as well. What makes this study so robust though is the rigour with which the centre documents all of their intubations using such a detailed registry. By using one centre much of the variability in practice between units is eliminated so perhaps these results can be trusted. Would your centre achieve these same results? Maybe not but it would certainly be interesting to test drive one of these for a period of time see how it performs.
by All Things Neonatal | Jul 26, 2018 | Neonatal, Neonatology, resuscitation
It is hard to believe but it has been almost 3 years since I wrote a piece entitled A 200 year old invention that remains king of all tech in newborn resuscitation. In the post I shared a recent story of a situation in which the EKG leads told a different story that what our ears and fingers would want us to believe. The concept of the piece was that in the setting of pulseless electrical activity (where there is electrical conductance in the myocardium but lack of contraction leaves no blood flow to the body) one could pick up a signal from the EKG leads when there is in fact no pulse or perfusion to vital organs. This single experience led me to postulate that this situation may be more common than we think and the application of EKG leads routinely could lead to errors in decision making during resuscitation of the newborn. It is easy to see how that could occur when you think about the racing pulses of our own in such situations and once chest compressions start one might watch the monitor and forget when they see a heart rate of 70 BPM to check for a corresponding pulse or listen with the stethoscope. I could see for example someone stopping chest compressions and continuing to provide BVM ventilation despite no palpable pulse when they see the QRS complex clearly on the monitor. I didn’t really have much evidence to support this concern but perhaps there is a little more to present now.
A Crafty Animal Study Provides The Evidence
I haven’t presented many animal studies but this one is fairly simple and serves to illustrate the concern in a research model. For those of you who haven’t done animal research, my apologies in advance as you read what happened to this group of piglets. Although it may sound awful, the study has demonstrated that the concern I and others have has is real.
For this study 54 newborn piglets (equivalent to 36-38 weeks GA in humans) were anesthetized and had a flow sensor surgically placed around the carotid artery. ECG leads were placed as well and then after achieving stabilization, hypoxia was induced with an FiO2 of 0.1 and then asphyxia by disconnecting the ventilator and clamping the ETT. By having a flow probe around the carotid artery the researchers were able to determine the point of no cardiac output and simultaneously monitor for electrical activity via the EKG leads. Auscultation for heart sounds was performed as well.
The results essentially confirm why I have been concerned with an over reliance on EKG leads.

Of the 57 piglets, 14 had asystole and no carotid flow but in 23 there was still a heart rate present on the EKG with no detectable carotid flow. This yields a sensitivity of only 37%. Moreover, the overall accuracy of the ECG was only 56%.
Meanwhile the stethoscope which I have referred to previously as the “king” in these situations had 100% sensitivity so remains deserving of that title.
What do we do with such information?
I think the results give us reason to pause and remember that faster isn’t always better. Previous research has shown that signal acquisition with EKG leads is faster than with oximetry. While a low heart rate detected quickly is helpful to know what the state of the infant is and begin the NRP pathway, we simply can’t rely on the EKG to tell us the whole story. We work in interdisciplinary teams and need to support one another in resuscitations and provide the team with the necessary information to perform well. The next time you are in such a situation remember that the EKG is only one part of the story and that auscultation for heart sounds and palpation of the umbilical cord for pulsation are necessary steps to demonstrate conclusively that you don’t just have a rhythm but a perfusing one.
I would like to thank the Edmonton group for continuing to put out such important work in the field of resuscitation!

by All Things Neonatal | Jun 30, 2018 | intubation, Neonatal, Neonatology, newborn, preemie, Prematurity
A few weeks back I wrote about the topic of intubations and whether premedication is really needed (Still performing awake intubations in newborns? Maybe this will change your mind.) I was clear in my belief that it is and offered reasons why. There is another group of practitioners though that generally agree that premedication is beneficial but have a different question. Many believe that analgesia or sedation is needed but question the need for paralysis. The usual argument is that if the intubation doesn’t go well and the patient can’t spontaneously ventilate could we be worse off if the patient loses their muscle tone.
Neonatal Intubation Registry
At the CPS meeting last month in Quebec City. I had the pleasure of listening to a talk by Dr. Elizabeth Foglia on the findings from a Neonatal intubation registry that many centres have been contributing to. The National Emergency Airway Registry for Neonates (NEAR4NEOs), records all intubations from a number of centres using an online database and allows for analysis of many different aspects of intubations in neonates.
This year, J. Krick et al published Premedication with paralysis improves intubation success and decreases adverse events in very low birth weight infants: a prospective cohort study. This study compared results from the registry of two centres, the University of Washington Medical Center (UWMC) and Seattle Children’s Hospital where the former rarely uses paralysis and the latter in almost all instances of non-emergent intubation. In all, 237 encounters were analyzed in the NICU for babies < 1500g with the majority of encounters (181) being from UWMC. The median PMA at intubation was 28 completed weeks (IQR: 27, 30), chronological age was 9 days (IQR: 2, 26) and weight was 953 g (IQR: 742,1200). The babies were compared based on the following groups. Premedication with a paralytic 21%, without a paralytic 46% and no premedication 31%.
This was an observational study that examined the rates of adverse events and subdivided into severe (cardiac arrest, esophageal intubation with delayed recognition, emesis with witnessed aspiration, hypotension requiring intervention (fluid and/or vasopressors), laryngospasm, malignant hyperthermia, pneumothorax/pneumomediastinum, or direct airway injury) vs non-severe (mainstem bronchial intuba- tion, esophageal intubation with immediate recognition, emesis without aspiration, hypertension requiring therapy, epistaxis, lip trauma, gum or oral trauma, dysrhythmia, and pain and/or agitation requiring additional medication and causing a delay in intubation.).
How did the groups compare?
It turns out paralysis seems to be a big deal (at least in this group of infants). Use of paralysis resulted in less attempts to intubate (median 1 attempt; IQR: 1, 2.25 vs. 2; IQR: 1, 3, p < 0.05)). In fact success was no different between the groups with no paralysis or no premedication at all! When it comes to tracheal intubation adverse events the impact of using paralysis becomes more evident. 
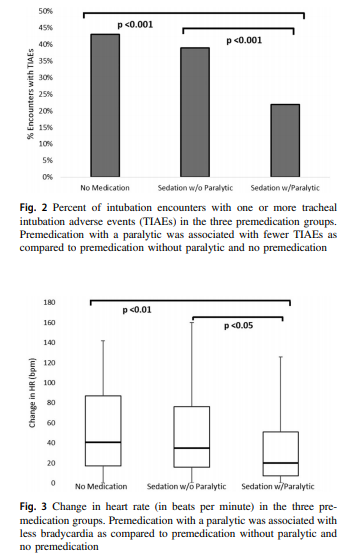 Paralysis does make a difference in reducing the incidence of such events and moreover when only looking at the rate of severe adverse events as defined above the finding was that none occurred when paralysis was used vs 9 when no paralysis was employed and 5 when no premedication was used at all. The rate of bradycardic events was less in the paralytic group but rates of oxygen desaturation between the three arms were no different.
Paralysis does make a difference in reducing the incidence of such events and moreover when only looking at the rate of severe adverse events as defined above the finding was that none occurred when paralysis was used vs 9 when no paralysis was employed and 5 when no premedication was used at all. The rate of bradycardic events was less in the paralytic group but rates of oxygen desaturation between the three arms were no different.
How do we interpret the results?
Based on the results from the registry it looks like paralysis is a good thing here when electively intubating infants. If we try to determine the reason for it I suspect it may have much to do with the higher likelihood of success on the first attempt at placing an ETT. The longer it takes to place the ETT or the more number of attempts requiring intermittent PPV in a patient who truly needs a tube the greater the likelihood that you will see adverse events including bradycardia. It may simply be that a calm and still patient is an easier intubation and getting the tube in faster yields a more stable patient.
I am biased though and I think it is worth pointing out another possible reason for the differing results. One hospital in this study routinely used premedication and the other did not. Almost 3/4 of the patients came from one hospital which raises the possibility that skill set could be playing a role. If the skill of providers at the two hospitals differed, the results could reflect the variable skill in the practitioners versus the difference in the medications used themselves. What I don’t know though is whether the two share the same training program or not. Are the trainees the same at both sites (google maps says the two sites are 11 minutes away by car)? The difference still might be in local respiratory therapists or Neonatologists intubating as well. Regardless, the study provides evidence that paralysis makes a difference. To convince those out there though who remain skeptical I think we are going to need the registry to take part in a prospective trial using many centres. A format in which several centres that don’t use paralysis are compared to several who do routinely would help to sort out the concern in skill when looking only at two centres. This wouldn’t be randomized of course but I think it would be very difficult at this point to get a centre that strongly believes in using paralysis to randomize so a prospective study using groups chosen by the individual centre might be the next best thing. If anyone using the registry is reading this let me know what you think?

by All Things Neonatal | Jun 27, 2018 | NAS, Neonatal, neonatal abstinence, Neonatology
 This post is very timely as the CPS Fetus and Newborn committee has just released a new practice point:
This post is very timely as the CPS Fetus and Newborn committee has just released a new practice point:
Managing infants born to mothers who have used opioids during pregnancy
Have a look at discharge considerations as that section in the statement speaks to this topic as well!
As bed pressures mount seemingly everywhere and “patient flow” becomes the catch-word of the day, wouldn’t it be nice to manage NAS patients in their homes? In many centres, such patients if hospitalized can take up to 3 weeks on average to discharge home off medications. Although done sporadically in our own centre, the question remains is one approach better than the another? Nothing is ever simple though and no doubt there are many factors to consider depending on where you live and what resources are available to you. Do you have outpatient follow-up at your disposal with practitioners well versed in the symptoms of NAS and moreover know what to do about them? Is there comfort in the first place with sending babies home on an opioid or phenobarbital with potential side effects of sedation and poor feeding? Nonetheless, the temptation to shift therapy from an inpatient to outpatient approach is very tempting.
The Tennessee Experience
Maalouf Fl et al have published an interesting account of the experience with outpatient therapy in their paper Outpatient Pharmacotherapy for Neonatal Abstinence Syndrome. The authors were able to take advantage of the Tennessee Medicaid program using administrative
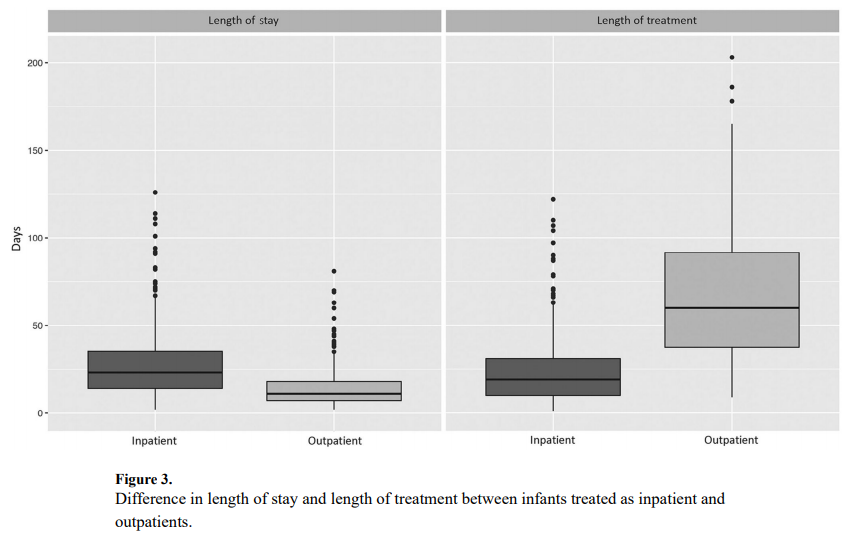
and vital records data from 2009 to 2011 to capture a cohort of 736 patients who were treated for NAS. Forty five percent or 242 patients were treated as outpatients vs 290 cared for in hospital for the duration of treatment. It is worth mentioning at this point that when the authors say they were cared for as outpatients it really is a hybrid model as the duration of hospitalization for the inpatients was a median of 23 days (IQR 14-35) versus 11 days (IQR 7-18) for inpatients (P < .001). This practice isn’t much different than my own in which I start therapy in hospital and then discharge home with a period of home therapy.
The strength of the study is the volume of patients and the ability to follow-up with these babies for the first 6 months of life to determine what happened to them after discharge. In terms of duration of treatment, the differences are significant but perhaps not surprising. The median length of treatment for outpatients was 60 days (IQR 38-92) compared with 19 days (IQR 10-31) for inpatients (P < .001). What was interesting as well is that 82% of babies were discharged home on phenobarbital and 9.1% on methadone and 7.4% with both. A very small minority was discharged home on something else such as morphine or clonidine. That there was a tripling of medication wean is not surprising as once the patients are out of the watchful eye of the medical team in hospital it is likely that practitioners would use a very slow wean out of hospital to minimize the risk of withdrawal.
An Unintended Consequence
This study found a statistically significant increase in risk for presenting to the emergency department for those patients treated as outpatients.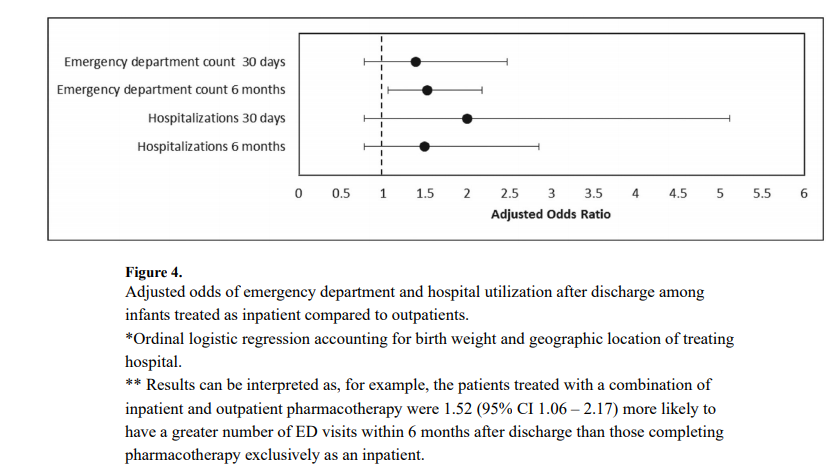

What this graph demonstrates is that there was no increase risk in the first month but there was for the first 6 months. Despite the increased risk of presentation to the ED the rate of hospitalization was not different. Drilling down the data further, the reason for coming to the ED was not for withdrawal which was 10% in the outpatient and 11% in the inpatient group. The other major reason was The most common diagnoses were upper respiratory infections; 80% outpatient vs 71% inpatient. So while there was a significant difference (which was not by much) my take on it is that it was most likely by chance as I can’t think of how infections in the first 6 months could be linked to choice of medication wean.
What about phenobarbital?
Phenobarbital has been used for many years in Neonatology for control of seizures, sedation (taking advantage of a side effect) and management of NAS. The problem with a median use of phenobarbital for 2 months is its potential to affect development.

An animal study by Diaz in 1999 in which rat pups were given two weeks of phenobarbital starting on day 5 of life and then euthanized demonstrated the following weight reductions when high dose phenobarbital was utilized. In human data, children with febrile seizures treated with phenobarbital in the paper Late cognitive effects of early treatment with phenobarbital. had decreased intelligence than those not exposed to phenobarbital.
The issue here for me is not necessarily whether babies can be treated successfully as outpatients for NAS. The concern is at what cost if the choice of drug is phenobarbital. The reason phenobarbital was chosen is likely due to compliance. We know that the more frequently a drug is dose the less likely compliance will be achieved. Phenobarbital being dosed either q12h or q24h is an ideal drug from a compliance point of view but the ramifications of this treatment deserve reconsideration.
I look forward to seeing further studies on this topic and hope that we see the results of an opioid outpatient treatment program. I know these exist and would welcome any information you as the readers of this blog can offer. Treating patients in the home makes great sense to me but we need to do it with the right drugs!

 In our new hospital we are grateful for the generosity of our Children’s Hospital Foundation who secured a donor to pay for a coordinator of such a program. The veteran parent who is leading this program ensures that no infant goes beyond a set period of time without feeling the touch or hearing the sound of a voice. Such a program is in fact already in place at our other tertiary hospital and was featured in a lovely article attached here. Taking all the information together that is out there I think that if we can provide the necessary stimulation from both touch and auditory stimuli as well we can provide these infants with the developmental needs that each of them requires.
In our new hospital we are grateful for the generosity of our Children’s Hospital Foundation who secured a donor to pay for a coordinator of such a program. The veteran parent who is leading this program ensures that no infant goes beyond a set period of time without feeling the touch or hearing the sound of a voice. Such a program is in fact already in place at our other tertiary hospital and was featured in a lovely article attached here. Taking all the information together that is out there I think that if we can provide the necessary stimulation from both touch and auditory stimuli as well we can provide these infants with the developmental needs that each of them requires.








 Paralysis does make a difference in reducing the incidence of such events and moreover when only looking at the rate of severe adverse events as defined above the finding was that none occurred when paralysis was used vs 9 when no paralysis was employed and 5 when no premedication was used at all. The rate of bradycardic events was less in the paralytic group but rates of oxygen desaturation between the three arms were no different.
Paralysis does make a difference in reducing the incidence of such events and moreover when only looking at the rate of severe adverse events as defined above the finding was that none occurred when paralysis was used vs 9 when no paralysis was employed and 5 when no premedication was used at all. The rate of bradycardic events was less in the paralytic group but rates of oxygen desaturation between the three arms were no different.
 This post is very timely as the CPS Fetus and Newborn committee has just released a new practice point:
This post is very timely as the CPS Fetus and Newborn committee has just released a new practice point:

