If you work in Neonatology then you can’t help but see babies with jaundice. It is so common in the NICU that I would call it normal. While mild jaundice is part of neonatal life, sometimes levels increase to the point that it can be dangerous to the developing brain and in the short term cause acute bilirubin encephalopathy. Chronic injury can lead to hearing impairment and kernicterus with staining of the basal ganglia and cerebral palsy. Phototherapy has been the mainstay of treatment with exchange transfusion reserved for exceptionally high levels of bilirubin. In recent years people have utilized IVIG when hemolysis is present to reduce the risk of exchange transfusion. There hasn’t been much else that one could use but many years ago, proto-porphyrins (SnPP)were looked at as a class of drug that might help reduce the burden of bilirubin by blocking where the substance comes from. Bilirubin is liberated when heme is broken down by heme-oxygenase so an inhibitor of this enzyme (of which the metallo-porphyrins (Sn-MP)might be the right candidate for the job. While initial trials seemed to show benefit there was a side effect of studies in rats indicated that exposure to Sn-PP (but not Sn-MP) led to photosensitivity if exposed to UV light and phototherapy. Given that the Sn-MP class of drugs didn’t seem to have the same issues an interest in this class of drugs has experienced a resurgence.
Phase IIb clinical trial
In the development of drugs there are different phases of trials. In a phase IIb trial, researchers believing from previous research that there is a treatment effect, perform a larger study using different dosing to see if there is still a benefit. In the case of Sn-MP this was done by Rosenfeld WN et al in their recent trial Stannsoporfin with phototherapy to treat hyperbilirubinemia in newborn hemolytic disease. The trial design included infants with hemolytic disease with 91 patients divided into three groups; 31 controls, 30 receiving a 3 mg/kg IM dose and 30 receiving a 4.5 mg/kg IM dose of stannsoporfin along with phototherapy. The goal of the study was to see whether infants receiving such treatments would experience different trajectories in their levels of bilirubin. In order to standardize things as much as possible, all infants were enrolled once they reached a pre-specified level using the AAP phototherapy curves and underwent serial TsB measurements at 2,6,12,24,36 and 48 hours after study entry.
Phototherapy was standardized based on measurement of spectral irradiance at the skin surface with maintenance of 30 microW/cm2/nm to ensure that each infant received the same level of phototherapy.
The Findings
I suppose it shouldn’t shock anyone that the drug works as the class of drug was found to work almost 40 years ago! Looking at the three groups over the 48 hour time frame shows a markedly different trajectory for the patients.
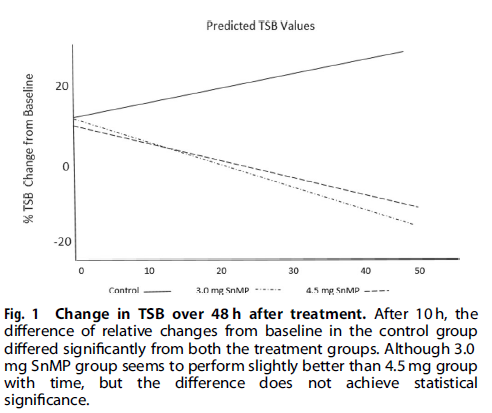
From a side effect standpoint, the following was noted:
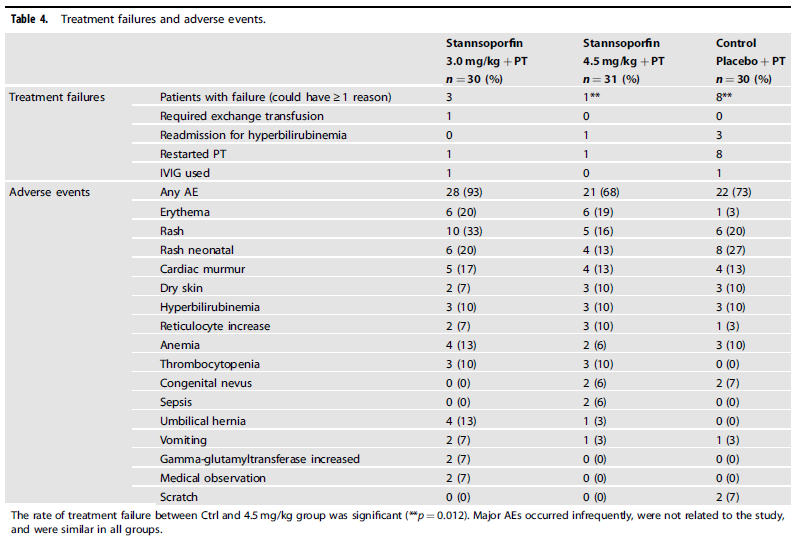
Is this ready for prime time?
The purpose of sharing this information with you is not to promote it as a treatment that we should all jump on. I think this story is still early. What will be needed from here are larger studies with well designed follow-up targeting long term outcomes. This study will be following up patients to see how they did but larger numbers will no doubt be wanted before people embrace this wholeheartedly. A quick search of trials on clinicaltrials.gov shows that there are several trials completed and no doubt some to come. Having an exchange transfusion is no laughing matter as there are risks related to clotting, bleeding, platelets etc so having another tool in our toolkit to deal with this common issue in neonates is of interest to me. Maybe it is of interest to you as well.


What is the half life of these drugs? Is it wise to use heme oxygenase inhibitors with a very long (years) half life? Perhaps this drug would be welcomed in certain Mediterranean populations, but do we really want to see this drug given to all newborns in the USA and Canada? I suspect if the drug were ever approved it would end up on most everyone’s standing order sets in the nursery. Count me out.