
by All Things Neonatal | Apr 1, 2018 | Neonatal, Neonatology, preemie, Prematurity, ventilation
For almost a decade now confirmation of intubation is to be done using detection of exhaled CO2. The 7th Edition of NRP has the following to say about confirmation of ETT placement “The primary methods of confirming endotracheal tube placement within the trachea are detecting exhaled CO2 and a rapidly rising heart rate.” They further acknowledge that there are two options for determining the presence of CO2 “There are 2 types of CO2 detectors available. Colorimetric devices change color in the presence of CO2. These are the most commonly used devices in the delivery room. Capnographs are electronic monitors that display the CO2 concentration with each breath.” The NRP program stops short of recommending one versus the other. I don’t have access to the costs of the colorimetric detectors but I would imagine they are MUCH cheaper than the equipment and sensors required to perform capnography using the NM3 monitor as an example. The real question though is if capnography is truly better and might change practice and create a safer resuscitation, is it the way to go?
Fast but not fast enough?
So we have a direct comparison to look at. Hunt KA st al published Detection of exhaled carbon dioxide following intubation during resuscitation at delivery this month. They started from the standpoint of knowing from the manufacturer of the Pedicap that it takes a partial pressure of CO2 of 4 mm Hg to begin seeing a colour change from purple to yellow but only when the CO2 reaches 15 mm Hg do you see a consistent colour change with that device. The capnograph from the NM3 monitor on the other hand is quantitative so is able to accurately display when those two thresholds are reached. This allowed the group to compare how long it took to see the first colour change compared to any detection of CO2 and then at the 4 and 15 mm Hg levels to see which is the quicker method of detection. It is an interesting question as what would happen if you were in a resuscitation and the person intubates and swears that they are in but there is no colour change for 5, 10 or 15 seconds or longer? At what point do you pull the ETT? Compare that with a quantitative method in which there is CO2 present but it is lower than 4. Would you leave the tube in and use more pressure (either PIP/PEEP or both?)? Before looking at the results, it will not shock you that ANY CO2 should be detected faster than two thresholds but does it make a difference to your resuscitation?
The Head to Head Comparison
The study was done retrospectively for 64 infants with a confirmed intubation using the NM3 monitor and capnography. Notably the centre did not use a colorimetric detector as a comparison group but rather relied on the manufacturers data indicating the 4 and 15 mm Hg thresholds for colour changes. The mean age of patients intubated was 27 weeks with a range of 23 – 34 weeks. The results I believe show something quite interesting and informative.
|
Median time secs (range) |
| Earliest CO2 detection |
3.7 (0 – 44s) |
| 4 mm Hg |
5.3 (0 – 727) |
| 15 mm Hg |
8.1 (0 – 727) |
I wouldn’t worry too much about a difference of 1.6 seconds to start getting a colour change but it is the range that has me a little worried. The vast majority of the patients demonstrated a level of 4 or 15 mm Hg within 50 seconds although many were found to take 25-50 seconds. When compared to a highest level of 44 seconds in the first detection of CO2 group it leads one to scratch their head. How many times have you been in a resuscitation and with no CO2 change you keep the ETT in past 25 seconds? Looking closer at the patients, there were 12 patients that took more than 30 seconds to reach a threshold of 4 mm Hg. All but one of the patients had a heart rate in between 60-85. Additionally there was an inverse relationship found between gestational age and time to detection. In other words, the smallest of the babies in the study took the longest to establish the threshold of 4 and 15 mm Hg.
Putting it into context?
What this study tells me is that the most fragile of infants may take the longest time to register a colour change using the colorimetric devices. It may well be that these infants take longer to open up their pulmonary vasculature and deliver CO2 to the alveoli. As well these same infants may take longer to open the lung and exhale the CO2. I suppose I worry that when a resuscitation is not going well and an infant at 25 weeks is bradycardic and being given PPV through an ETT without colour change, are they really not intubated? In our own centre we use capnometry in these infants (looks for a wave form of CO2) which may be the best option if you are looking to avoid purchasing equipment for quantitative CO2 measurements. I do worry though that in places where the colorimetric devices are used for all there will be patients who are extubated due to the thought that they in fact have an esophageal intubation when the truth is they just need time to get the CO2 high enough to register a change in colour.
Anyways, this is food for thought and a chance to look at your own practice and see if it is in need of a tweak…

by All Things Neonatal | Mar 14, 2018 | apnea of prematurity, caffeine, extubation, Neonatal, Neonatology, Prematurity, ventilation
Caffeine seems to be good for preterm infants. We know that it reduces the frequency of apnea in the this population and moreover facilitates weaning off the ventilator in a shorter time frame than if one never received it at all. The earlier you give it also seems to make a difference as shown in the Cochrane review on prophylactic caffeine. When given in such a fashion the chances of successful extubation increase. Less time on the ventilator not surprisingly leads to less chronic lung disease which is also a good thing.
I have written about caffeine more than once though so why is this post different? The question now seems to be how much caffeine is enough to get the best outcomes for our infants. Last month I wrote about the fact that as the half life of caffeine in the growing preterm infant shortens, our strategy in the NICU might be to change the dosing of caffeine as the patient ages. Some time ago though I wrote about the use of higher doses of caffeine and in the study analyzed warned that there had been a finding of increased cerebellar hemorrhage in the group randomized to receive the higher dosing. I don’t know about where you work but we are starting to see a trend towards using higher caffeine base dosing above 5 mg/kg/d. Essentially, we are at times “titrating to effect” with dosing being as high as 8-10 mg/kg/d of caffeine base.
Does it work to improve meaningful outcomes?
This month Vliegenthart R et al published a systematic review of all RCTs that compared a high vs low dosing strategy for caffeine in infants under 32 weeks at birth; High versus standard dose caffeine for apnoea: a systematic review. All told there were 6 studies that met the criteria for inclusion. Low dosing (all in caffeine base) was considered to be 5- 15 mg/kg with a maintenance dose of 2.5 mg/kg to 5 mg/kg. High dosing was a load of 5 mg/kg to 40 mg/kg with a maintenance of 2.5 mg/kg to 15 mg/kg. The variability in the dosing (some of which I would not consider high at all) makes the quality of the included studies questionable so a word of warning that the results may not truly be “high” vs “low” but rather “inconsistently high” vs. “inconsistently low”.
The results though may show some interesting findings that I think provide some reassurance that higher dosing can allow us to sleep at night.
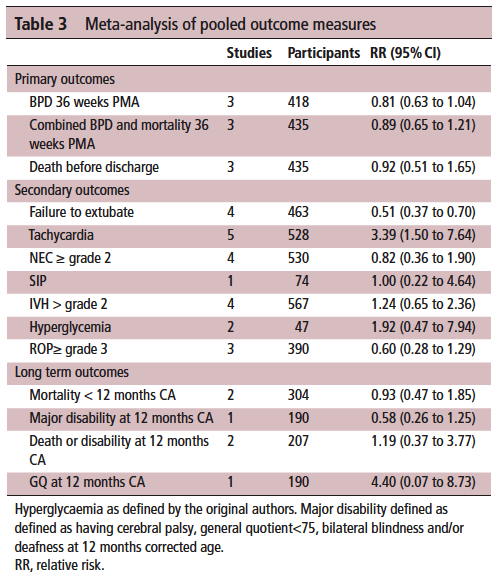
On the positive front, while there was no benefit to BPD and mortality at 36 weeks PMA they did find if they looked only at those babies who were treated with caffeine greater than 14 days there was a statistically significant difference in both reduction of BPD and decreased risk of BPD and mortality. This makes quite a bit of sense if you think about it for a moment. If we know that caffeine improves the chances of successful extubation and we also know it reduces apnea, then who might be on caffeine for less than 2 weeks? The most stable of babies I would expect! These babies were all < 32 weeks at birth. What the review suggests is that those babies who needed caffeine for longer durations benefit the most from the higher dose. I think I can buy that.
On the adverse event side, I suppose it shouldn’t surprise many that the risk of tachycardia was statistically increased with an RR of 3.4. Anyone who has explored higher dosing would certainly buy that as a side effect that we probably didn’t need an RCT to prove to us. Never mind that, have you ever taken your own pulse after a couple strong coffees in the morning?
What did it not show?
It’s what the study didn’t show that is almost equally interesting. The cerebellar hemorrhages seen in the study I previously wrote about were not seen at all in the other studies. There could be a lesson in there about taking too much stock in secondary outcomes in small studies…
Also of note, looking at longer term outcome measures there appears to be no evidence of harm when the patients are all pooled together. The total number of patients in all of these studies was 620 which for a neonatal systematic review is not bad. A larger RCT may be needed to conclusively tell us what to do with a high and low dosing strategy that we can all agree on. What do we do though in the here and now? More specifically, if you are on call tomorrow and a baby is on 5 mg/kg/d of caffeine already, will you intubate them if they are having copious apneic events or give them a higher dose of caffeine when CPAP or NIPPV that they are already on isn’t cutting it? That is where the truth about how you feel about the evidence really comes out. These decisions are never easy but unfortunately you sometimes have to make a decision and the perfect study hasn’t been done yet. I am not sure where you sit on this but I think this study while certainly flawed gives me some comfort that nothing is truly standing out especially given the fact that some of the “high dose” studies were truly high. Will see what happens with my next patient!
by All Things Neonatal | Jan 24, 2018 | caffeine, Neonatal, Neonatology, preemie, Prematurity
This has been a question that has befuddled Neonatologists for years. Get ten of us in a room and you will get a variety of responses ranging from (talking about caffeine base) 2.5 mg/kg/day to 10 mg/kg/day. We will espouse all of our reasons and question the issue of safety at higher doses but in the end do we really know? As I was speaking to a colleague in Calgary yesterday we talked about how convinced we are of our current management strategies but how we both recognize that half of what we think we know today we will be questioning in 10 years. So how convinced should we really be about caffeine?
Even the Cochrane Review Suggests There Is Something Amiss
Back in 2010 the Cochrane Collaboration examining 6 trials on caffeine for treating apnea of prematurity concluded “Methylxanthine is effective in reducing the number of apnoeic attacks and the use of mechanical ventilation in the two to seven days after starting treatment.” Notice the bolded section. Two to seven days. Interesting that we don’t see the effect last in perpetuity. Why might that be? Do babies become resistant with time or is there a change in the way these infants metabolize the drug such that levels in the bloodstream drop after that time point. It is almost certainly the latter and in the last 7 years have we really seen any response to this finding? I would say no for the most part although I don’t work in your unit so hard to say for sure. At least where I practice we pick a dose somewhere between 2.5-5 mg/kg/day and give a load of 10 mg/kg when we start the drug. From time to time we give a miniload of 5 mg/kg and may or may not increase the dose of maintenance based on the number of apneic events the babies are having. What if we could be proactive instead of reactive though. Do the babies need to have multiple events before we act or could we prevent the events from happening at all?
Proactive Treatment With Caffeine
We have known that caffeine clearance increases with postnatal age. The half-life of the drug shortens from about a week at the earliest gestational ages to 2-2.5 days by term equivalent age. For those infants who are older such as 32 weeks and above we expect them to be off caffeine (if they need it) within 2-3 weeks so I am not really talking about them but what about the babies born earlier than that or certainly MUCH earlier at 23 and 24 weeks who will be on caffeine possibly till term. Should one size (dose) fit all? No it really shouldn’t and some crafty researchers led by Koch G have published a paper that demonstrates why entitled Caffeine Citrate Dosing Adjustments to Assure Stable Caffeine Concentrations in Preterm Neonates.
In this paper the authors armed with knowledge of the half life of caffeine at different gestational ages were able to calculate the clearance of the drug at different postnatal ages to demonstrate in a model of a 28 week male infant weighing 1150g. The authors further took into account predicted weight changes and were able to calculate what the expected caffeine levels would be in the fictional infant at various time points. The target caffeine levels for this patient were a trough level of 15 -20 mg/L which are the currently acceptable ranges in the literature. The testing was first done using a standard load of 10 mg/kg (base) followed by 2.5 mg/kg/d (base) and demonstrated levels which yielded the following graph over time. 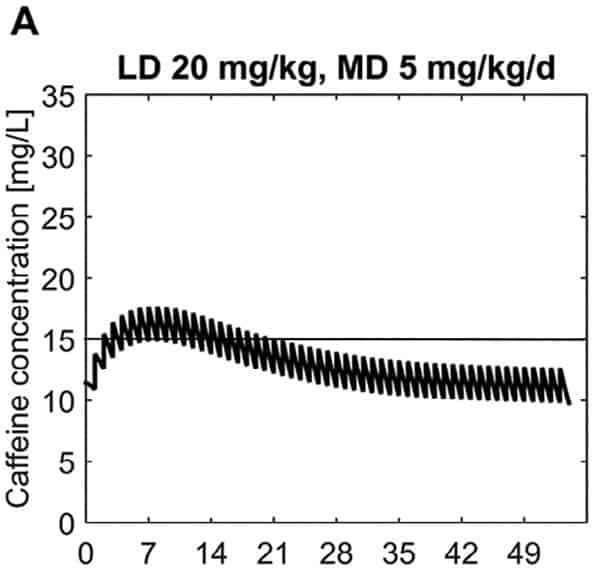 What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next.
What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next. 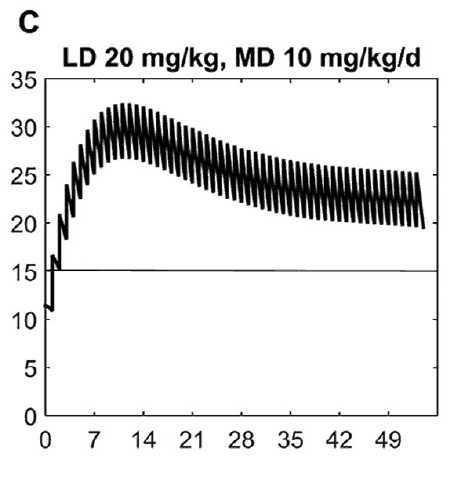 Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve.
Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve.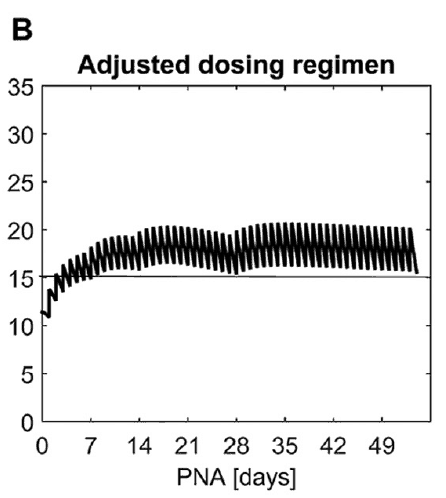 As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
A Suggestion For The Future
What grabbed my attention here is the possibility that we could take a proactive rather than reactive approach to these infants. Once a small baby is controlled on their dose of caffeine whether it is 2.5, 3, 5 or even 6 mg/kg/d of caffeine should we wait for more events to occur and then react by increasing caffeine? What if we are too late to respond and the patient is intubated. What effect does this have on the developing lung, what about the brain that is subjected to bradycardic events with resultant drops in cardiac output and cerebral perfusion. Perhaps the solution is to work with our pharmacists and plan to increase dosing at several time points in the infants journey through the NICU even if they aren’t showing symptoms yet. No doubt this is a change in approach at least for the unit I work in but one that should start with a conversation!
by All Things Neonatal | Jan 15, 2018 | BPD, Neonatal, Neonatology, newborn, outcome, preemie, Prematurity, steroids
This must be one of my favourite topics as I have been following the story of early hydrocortisone to reduce BPD for quite some time. It becomes even more enticing when I have met the authors of the studies previously and can see how passionate they are about the possibilities. The PREMILOC study was covered on my site twice now, with the first post being A Shocking Change in Position. Postnatal steroids for ALL microprems? and the second reviewing the 22 month outcome afterwards /2017/05/07/early-hydrocortisone-short-term-gain-without-long-term-pain/.
The intervention here was that within 24 hours of birth babies born between 24-27 weeks gestational age were randomized to receive placebo or hydrocortisone 1 mg/kg/d divided q12h for one week followed by 0.5 mg/kg/d for three days. The primary outcome was rate of survival without BPD at 36 weeks PMA. The finding was a positive one with a 9% reduction in this outcome with the use of this strategy. Following these results were the two year follow-up which reported no evidence of harm but the planned analysis by gestational age groupings of 24-25 and 26-27 weeks was not reported at that time but it has just been released this month.
Is there a benefit?
Of the original cohort the authors are to be commended here as they were able to follow-up 93% of all infants studied at a mean age of 22 months. The methods of assessing their neurological status have been discussed previously but essentially comprised standardized questionnaires for parents, assessment tools and physical examinations.
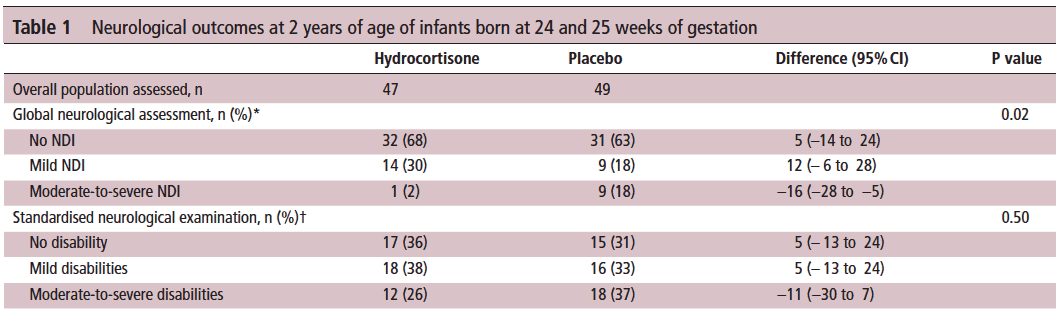
Let’s start off with what they didn’t find. There was no difference between those who received placebo vs hydrocortisone in the 26-27 week group but where it perhaps matters most there was. The infants born at 24-25 weeks are certainly some of our highest risk infants in the NICU. It is in this group that the use of hydrocortisone translated into a statistically significant reduction in the rate of neurodevelopmental impairment. The Global Neurological Assessement scores demonstrated a significant improvement in the hydrocortisone group with a p value of 0.02. Specifically moderate to severe disability was noted in 18% compared to 2% in the group receiving hydrocortisone.They did not find a difference in the neurological exam but that may reflect the lack of physical abnormalities with cognitive deficit remaining. It could also be explained perhaps by the physical examination not being sensitive enough to capture subtle differences.
Why might this be?
Adding an anti-inflammatory agent into the early phase of a preemies life might spare the brain from white matter damage. Inflammation is well known to inflict injury upon the developing brain and other organs (think BPD, ROP) so dampening these factors in the first ten days of life could bring about such results via a mechanism such as that. When you look at the original findings of the study though, a couple other factors also pop up that likely contribute to these findings as well. Infants in the hydrocortisone group had a statistical reduction in the rate of BPD and PDA ligations. Both of these outcomes have been independently linked to adverse neurodevelopmental outcome so it stands to reason that reducing each of these outcomes in the most vulnerable infants could have a benefit.
In fact when you add everything up, is there much reason not to try this approach? Ten days of hydrocortisone has now been shown to reduce BPD, decrease PDA ligations and importantly in the most vulnerable of our infants improve their developmental outcome. I think with this information at our fingertips it becomes increasingly difficult to ignore this approach. Do I think this will become adopted widely? I suspect there will be those who take the Cochrane approach to this and will ask for more well designed RCTs to be done in order to replicate these results or at least confirm a direction of effect which can then be studied as part of a systematic review. There will be those early adopters though who may well take this on. It will be interesting to see as these centres in turn report their before and after comparisons in the literature what the real world impact of this approach might be.
Stay tuned as I am sure this is not the last we will hear on this topic!
by All Things Neonatal | Dec 28, 2017 | preemie, Prematurity, technology, ventilation
Intubation is not an easy skill to maintain with the declining opportunities that exist as we move more and more to supporting neonates with CPAP. In the tertiary centres this is true and even more so in rural centres or non academic sites where the number of deliveries are lower and the number of infants born before 37 weeks gestational age even smaller. If you are a practitioner working in such a centre you may relate to the following scenario. A woman comes in unexpectedly at 33 weeks gestational age and is in active labour. She is assessed and found to be 8 cm and is too far along to transport. The provider calls for support but there will be an estimated two hours for a team to arrive to retrieve the infant who is about to be born. The baby is born 30 minutes later and develops significant respiratory distress. There is a t-piece resuscitator available but despite application the baby needs 40% oxygen and continues to work hard to breathe. A call is made to the transport team who asks if you can intubate and give surfactant. Your reply is that you haven’t intubated in quite some time and aren’t sure if you can do it. It is in this scenario that the following strategy might be helpful.
Surfactant Administration Through and Laryngeal Mask Airway (LMA)
Use of an LMA has been taught for years in NRP now as a good choice to support ventilation when one can’t intubate. The device is easy enough to insert and given that it has a central lumen through which gases are exchanged it provides a means by which surfactant could be instilled through a catheter placed down the lumen of the device. Roberts KD et al published an interesting unmasked but randomized study on this topic Laryngeal Mask Airway for Surfactant Administration in Neonates: A Randomized, Controlled Trial. Due to size limitations (ELBWs are too small to use this in using LMA devices) the eligible infants included those from 28 0/7 to 35 6/7 weeks and ≥1250 g. The infants needed to all be on CPAP +6 first and then fell into one of two treatment groups based on the following inclusion criteria: age ≤36 hours,
(FiO2) 0.30-0.40 for ≥30 minutes (target SpO2 88% and 92%), and chest radiograph and clinical presentation consistent with RDS.
Exclusion criteria included prior mechanical ventilation or surfactant administration, major congenital anomalies, abnormality of the airway, respiratory distress because of an etiology other than RDS, or an Apgar score <5 at 5 minutes of age.
Procedure & Primary Outcome
After the LMA was placed a y-connector was attached to the proximal end. On one side a CO2 detector was placed and then a bag valve mask in order to provide manual breaths and confirm placement over the airway. The other port was used to advance a catheter and administer curosurf in 2 mL aliquots. Prior to and then at the conclusion of the procedure the stomach contents were aspirated and the amount of surfactant determined to provide an estimate of how much surfactant was delivered to the lungs. The primary outcome was treatment failure necessitating intubation and mechanical ventilation in the first 7 days of life. Treatment failure was defined upfront and required 2 of the following: (1) FiO2 >0.40 for >30
minutes (to maintain SpO2 between 88% and 92%), (2) PCO2 >65 mmHg on arterial or capillary blood gas or >70 on venous blood gas, or (3) pH <7.22 or 1 of the following: (1) recurrent or severe apnea, (2) hemodynamic instability requiring pressors, (3) repeat surfactant dose, or (4) deemed necessary by medical provider.
Did it work?
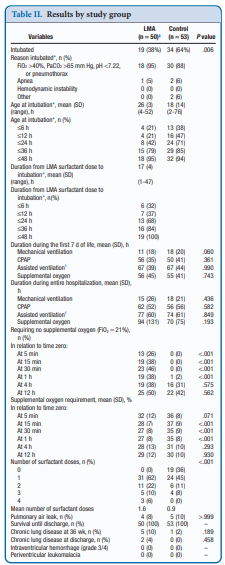
It actually did. Of the 103 patients enrolled (50 LMA and 53 control) 38% required intubation in the LMA group vs 64% in the control arm. The authors did not reach their desired enrollment based on their power calculation but that is ok given that they found a difference. What is really interesting is that they found a difference in the clinical end point despite many infants clearly not receiving a full dose of surfactant as measured by gastric aspirate. Roughly 25% of the infants were found to have not received any surfactant, 20% had >50% of the dose in the stomach and the other 50+% had < 10% of the dose in the stomach meaning that the majority was in fact deposited in the lungs. I suppose it shouldn’t come as a surprise that among the secondary outcomes the duration length of mechanical ventilation did not differ between two groups which I presume occurred due to the babies needing intubation being similar. If you needed it you needed it so to speak. Further evidence though of the effectiveness of the therapy was that the average FiO2 30 minutes after being treated was significantly lower in the group with the LMA treatment 27 vs 35%. What would have been interesting to see is if you excluded the patients who received little or no surfactant, how did the ones treated with intratracheal deposition of the dose fare? One nice thing to see though was the lack of harm as evidenced by no increased rate of pneumothorax, prolonged ventilation or higher oxygen.
Should we do this routinely?
There was a 26% reduction in intubations in te LMA group which if we take this as the absolute risk reduction means that for every 4 patients treated with an LMA surfactant approach, one patient will avoid intubation. That is pretty darn good! If we also take into account that in the real world, if we thought that little of the surfactant entered the lung we would reapply the mask and try the treatment again. Even if we didn’t do it right away we might do it hours later.
In a tertiary care centre, this approach may not be needed as a primary method. If you fail to intubate though for surfactant this might well be a safe approach to try while waiting for a more definitive airway. Importantly this won’t help you below 28 weeks or 1250g as the LMA is too small but with smaller LMAs might this be possible. Stay tuned as I suspect this is not the last we will hear of this strategy!
by All Things Neonatal | Nov 16, 2017 | Neonatal, Neonatology, preemie, Prematurity
Given that today is world prematurity day it seems fitting to talk about prematurity at the absolute extreme of it.
It has been some time since as a regional program we came to accept that we would offer resuscitation to preterm infants born as early as 23 weeks gestational age. This is perhaps a little later in the game that other centers but it took time to digest the idea that the rate of intact survival was high enough to warrant a trial of resuscitation. This of course is not a unilateral decision but rather a decision arrived at after consultation with the family and interprofessional team. To be sure it is not an easy one. Other centers have argued that resuscitation should be offered to those infants as young as 22 weeks gestational age and data now exists due to enough centres doing so to provide families with some guidance as to expected survival rates and importantly the likelihood of disability. This topic has been covered previously in /2015/09/25/winnipeg-hospital-about-to-start-resuscitating-infants-at-23-weeks/. Why cover this topic again? Well an article on CNN might have something to do with it.
Resuscitating Below 22 weeks
This week as I was perusing the news I came across a rather shocking article on CNN. Born before 22 weeks, ‘most premature’ baby is now thriving.  The article tells the tale of a baby delivered at 21 weeks and 4 days that now as a three year old is reaching appropriate milestones without any significant impairments. It is a story that is filled with inspiration and so I am not mistaken I am delighted for this child and their family that this outcome has occurred. When the lay press latches onto stories like this there is no doubt a great deal of sensationalism to them and in turn that gathers a lot of attention. This in turn is a great thing for media.
The article tells the tale of a baby delivered at 21 weeks and 4 days that now as a three year old is reaching appropriate milestones without any significant impairments. It is a story that is filled with inspiration and so I am not mistaken I am delighted for this child and their family that this outcome has occurred. When the lay press latches onto stories like this there is no doubt a great deal of sensationalism to them and in turn that gathers a lot of attention. This in turn is a great thing for media.
A Few Caveats Though
With the exception of pregnancies conceived through IVF the best dating we have is only good to about +/- 5 days when an early first trimester ultrasound is performed or the date of the last menstrual period is fairly certain. A baby though who is born at 21 weeks + 4 days may in fact be 22 +3 days or even more depending on when the dating was done (second trimester worse). Let’s not take away though from the outcome being this good even at 22 weeks. That is a pretty perfect outcome for this family but the point is that this baby may in fact be older than 21 weeks.
Secondly, there are millions of babies born each year in North America. Some of these infants are born at 22 weeks. How do they fare overall? From the paper by Rysavy et al from 2015 the results are as follows.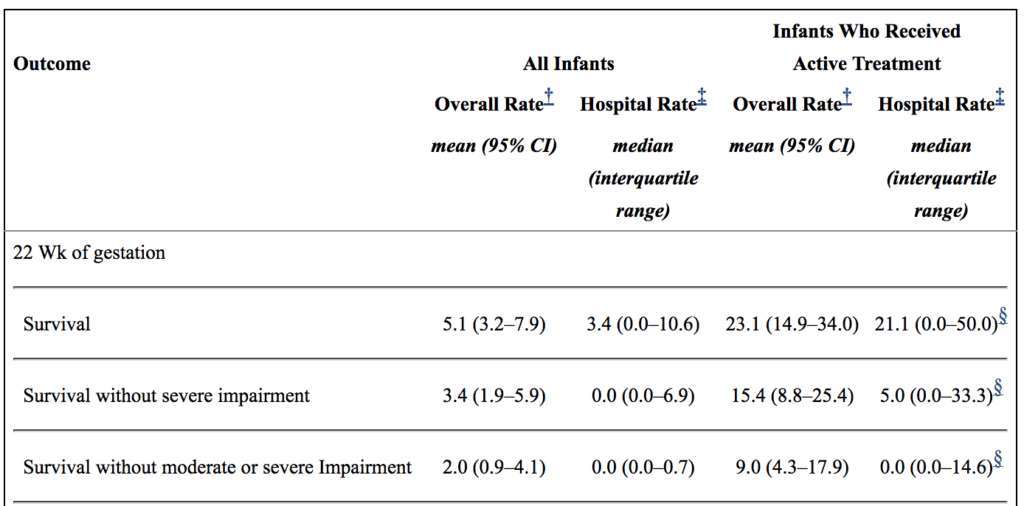
If you look at the overall rate of survival it is on an average of 5.1%. If you take a look though at those infants in whom resuscitation is provided that number increases to a mean of 23%. Intact survival is 9% overall. The odds aren’t great but they are there and I suspect the infant in the article is one of those babies. Flipping the argument though to the glass is half empty, 91% of infants born at 22 weeks by best estimate who are offered resuscitation will have a moderate or severe disability or die. I am not saying what one should do in this situation but depending on how a family processes the data they will either see the 110 chance of intact survival as a good thing or a 9/10 chance of death or disability as a very bad thing. What a family chooses though is anyone’s best guess.
Should we resuscitate below 22 weeks if the family wishes?
I guess in the end this really depends on a couple things. First off, how certain are the dates? If there is any degree of uncertainty then perhaps the answer is yes. If the dates are firm then I at least believe there is a barrier at which futility is reached. Perhaps this isn’t at 21 weeks as some patients may indeed be older but think about what you would offer if a family presented at 20 weeks and wanted everything done. What if it were 19 weeks? I suspect the point of futility for all lies somewhere between 19-21 weeks.
As I prepare to attend the annual meeting in Ottawa tomorrow for the Fetus and Newborn Committee I think it is prudent to point out just how difficult all of this is. The current statement on Counselling and management for anticipated extremely preterm birth I think hits on many of these issues. The statement is the product on not only the think tank that exists on this committee but was the product of a national consultation. I know I may be biased since I sit on the committee but I do believe it really hits the mark.
Should we be thinking about resuscitating at 21 weeks? For me the answer is one clouded by a whole host of variables and not one that can be easily answered here. What I do think though is that the answer in the future may be a yes provided such infants can be put onto an artificial placenta.  Even getting a few more weeks of growth before aerating those lungs is necessary may make all the difference. The NICUs of tomorrow certainly may look quite different than they do now.
Even getting a few more weeks of growth before aerating those lungs is necessary may make all the difference. The NICUs of tomorrow certainly may look quite different than they do now.





 What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next.
What this demonstrates is that if the dose is unchanged over the first 7 weeks, this hypothetical infant will only achieve effective concentrations for the first week. Interesting isn’t it that the Cochrane review found clinical effect over the first 2-7 days? What if you were to double the dose to really “hit” the infant with a good dose of caffeine from the start and maintain at that level based on their weight gain as shown next.  Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve.
Well, you will get what you are hoping for and keep the trough level above 15 mg/L but you will hit 30 mg/L that some have said is too high and can lead to adverse effects (ever seen SVT with these high doses? I have). Like Goldilocks and the Three Bears could there be a dosing strategy that might be just right? The authors put in another model based on the knowledge of caffeine clearance over time and suggested a strategy in which after the first week the adjusted maintenance doses would be 3 mg/kg/day and 3.5 mg/kg/day in the third to fourth weeks and lastly 4 mg/kg/d in the 5th to 8th week. Using that dosing schedule the model produced this curve. As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.
As you can see, the infant would have a therapeutic target without reaching levels above 30 mg/L and potential for side effects. As many of you read this however you may ask the obvious question. Each of us have seen infants who require higher doses than this to rid themselves of significant apnea and escape reintubation. Given that this is a mathematical model it assumes that this fictional infant will respond beautifully to a trough level of 15 to 20 mg/L but some will not. Even in the curve shown it is clear that there is some room to go higher in the dosing as the curve is just touching 20 mg/L.




 The article tells the tale of a baby delivered at 21 weeks and 4 days that now as a three year old is reaching appropriate milestones without any significant impairments. It is a story that is filled with inspiration and so I am not mistaken I am delighted for this child and their family that this outcome has occurred. When the lay press latches onto stories like this there is no doubt a great deal of sensationalism to them and in turn that gathers a lot of attention. This in turn is a great thing for media.
The article tells the tale of a baby delivered at 21 weeks and 4 days that now as a three year old is reaching appropriate milestones without any significant impairments. It is a story that is filled with inspiration and so I am not mistaken I am delighted for this child and their family that this outcome has occurred. When the lay press latches onto stories like this there is no doubt a great deal of sensationalism to them and in turn that gathers a lot of attention. This in turn is a great thing for media.
 Even getting a few more weeks of growth before aerating those lungs is necessary may make all the difference. The NICUs of tomorrow certainly may look quite different than they do now.
Even getting a few more weeks of growth before aerating those lungs is necessary may make all the difference. The NICUs of tomorrow certainly may look quite different than they do now.